Muhammad Saiful Islam, Mohammad Akhtar Hussain, Peter Black, and Andrea Britton
Nipah virus (NiV) is an emerging zoonotic pathogen from the same virus family as Hendra virus, already found in Australia, which is causing severe infectious disease in parts of the Indo-Pacific region.
NiV was first identified during outbreaks of encephalitis in Malaysia and Singapore between October 1998 to June 1999 and was associated with a spillover of the virus from bats into pigs, and then humans.
Although no further outbreaks have been reported in Malaysia and Singapore since, India, Bangladesh, and the Philippines have reported sporadic clusters of NiV infection. In the past 25 years Bangladesh alone has documented outbreaks of roughly 14 cases every year.
Recent WHO reports of NiV infection in healthcare workers in India and a community member in Bangladesh in early 2026 have drawn renewed attention to the virus. NiV is particularly concerning not only because of its high case fatality rate and the absence of licensed vaccines or therapeutics, but because of its capacity for sustained human-to-human transmission, including documented multi-generational transmission chains.
While the risk of infection to most Australians remains extremely low, understanding NiV, its transmission pathways, transmissibility, and prevention strategies is important for travellers to affected regions, and for broader biosecurity and epidemic preparedness.
What is Nipah virus?
NiV is an emerging pathogen, with Pteropus spp. bats the natural reservoir. The genetic diversity of this bat virus is not well understood. It is highly pathogenic, enveloped, single-stranded RNA virus belonging to new genus Henipavirus, of the family Paramyxoviridae.
Case fatality rate of NiV can reach up to 80%, while up to 60% of NiV survivors have neurological symptoms due to encephalitis. There are two recognised genotypes, NiV-M and NiV-B, that appear to differ slightly epidemiologically.
How it spreads to animals and humans
NiV transmission is associated with the interface of bat ecology, food systems, health care practices, and family caregiving practices. Spillover to domestic animals such as pigs can occur through the consumption of food contaminated with bat saliva, urine, or faeces. Evidence of NiV exposure has also been detected in cattle, dogs, and cats in Bangladesh.
Human infection most commonly occurs through consumption of raw or partially fermented date palm sap contaminated by bat saliva, urine or faeces. However, exposure to partially eaten fruit, and direct contact with infected animals have also been documented as important risk pathways.
Human-to-human transmission is well-documented and occurs primarily through direct contact with infected individuals or their body secretions, with health-care workers at greatest risk.
Pandemic potential of Nipah virus: lesson from past outbreaks
Early NiV outbreaks in Malaysia and Singapore were largely driven by an intermediate host (pigs), with limited evidence of person-to-person transmission.
That history occasionally created a false sense of reassurance that “NiV spreads poorly between people.” Outbreaks in Bangladesh and India have however repeatedly challenged that assumption. In Siliguri, India, a large proportion of cases in 2001 were linked to hospital exposure, including many healthcare workers. Several outbreaks in Bangladesh have shown increased risk to people who lived with, cared for, or had close contact with symptomatic patients.
The most compelling evidence comes from the 2024 outbreak in Bangladesh, where detailed contact tracing documented sustained person-to-person transmission extending to the fifth generation.
A community leader was linked to large number of subsequent infections, illustrating a superspreading event now familiar from other epidemic-prone pathogens. While not every case drives transmission, certain roles and care patterns can lead to increased risk of transmission. This finding is critical for preparedness.
Fifth-generation transmission demonstrates that NiV can move beyond short, self-limiting clusters under favourable conditions — particularly where close caregiving, crowding, delayed diagnosis, and limited infection prevention and control capacity are evident.
The 2026 cases in India and Bangladesh reflect these same infected bats to palm sap to human-to-human transmission pathways. In West Bengal (India), infections among healthcare workers in the same private hospital prompted extensive contact tracing and strengthened infection prevention and control measures, highlighting how healthcare facilities can act as amplifiers when early symptoms are non-specific.
In Bangladesh, the 2026 case again underscores the role of foodborne exposure linked to raw virus-contaminated date palm sap, highlighting a One Health spillover pathway of key concern where bat ecology intersects with cultural food practices.
Why this matters for Australia
Although the current risk of NiV introduction into Australia is very low, incursion is theoretically possible through two pathways: bat reservoirs and international travel. Pteropus fruit bats, the natural reservoir of NiV, have a broad geographic distribution extending from Africa to South and Southeast Asia into Australia. While NiV has not been detected in Australian flying foxes, closely related henipaviruses such as Hendra virus are endemic.
Evidence of NiV infection has been identified in bat populations in several of Australia’s nearest neighbours, including Papua New Guinea, Timor Leste, Indonesia. Qualitative risk assessments suggest that establishment of NiV in Australian flying foxes through pre-border bat movements is unlikely, though continued surveillance is recommended for early detection.
International travel represents a second pathway, with some countries such as Thailand and India having introduced airport temperature screening.
NiV infection typically causes severe illness that may limit travel; however, the incubation period ranges from approximately three to 14 days or longer, creating a window in which an exposed but asymptomatic person could travel internationally.
These pathways underscore why NiV remains relevant to Australia’s pandemic preparedness, highlighting the need for a coordinated One Health approach with strong surveillance, and rapid early detection of animal and/or human exposures to prevent outbreaks.
Dr Md Saiful Islam1, Dr Mohammad Akhtar Hussain2, Dr Peter Black3, and Dr Andrea Britton4
The authors are members of the Public Health Association of Australia’s One Health Special Interest Group, of which Dr Black and Dr Britton are co-convenors.
1School of Population Health, University of New South Wales, Sydney, Australia
2Barwon South West Public Health Unit, Geelong, Victoria
3Centre for Environment and Population Health, Griffith University
4One Health Advisor and Director, Ultimate Efficacy Consulting Ltd
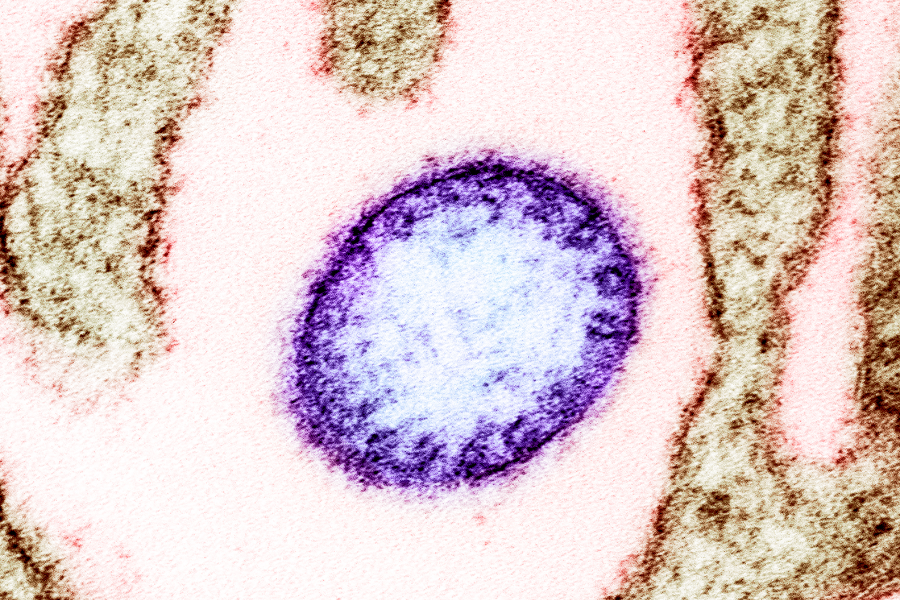
Image: Colorized transmission electron micrograph of a mature extracellular Nipah Virus particle (purple) near the periphery of an infected VERO cell (brown). Image captured and color-enhanced at the NIAID Integrated Research Facility in Fort Detrick, Maryland. Credit: NIAID


Leave a Reply